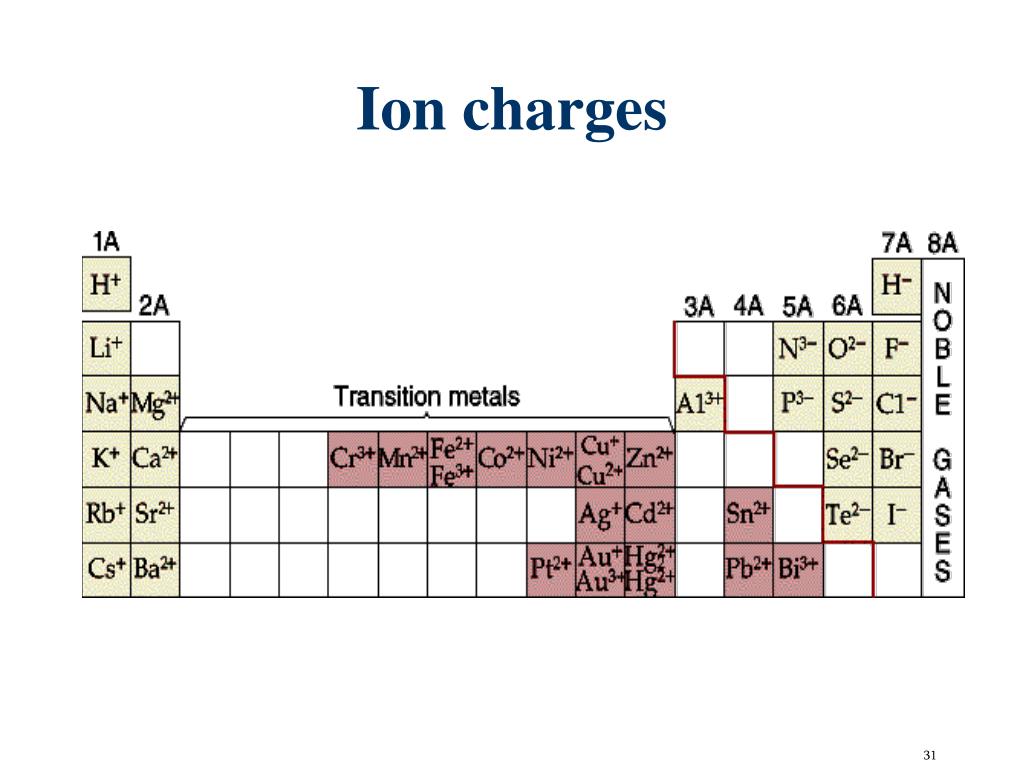 
Use a periodic table to determine the charges achieved upon ionization of main group elements. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. We begin with the elements in their most common states, Cs( s) and F 2( g). State the charge pattern for main group element ionization. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. Ions are also deflected by a magnetic field, as you saw in the opening image of the northern lights. Ions with the same charge, on the other hand, repel each other. This occurs because oppositely charged ions attract each other. When sodium and chlorine come into contact, each sodium atom gives up an electron to become a Na + ion, with 11 protons in its nucleus but only 10 electrons (like neon), and each chlorine atom gains an electron to become a Cl ion, with 17 protons in its nucleus and 18 electrons (like argon), as shown in part (b) in Figure 4.2.3.1 4.2.3. \): The Born-Haber cycle shows the relative energies of each step involved in the formation of an ionic solid from the necessary elements in their reference states. Periodic Table of the Elements Chemistry Reference Sheet California Standards Test 11 Na Sodium 22.99 Element symbol Element name 1 H Hydrogen 1.01 3 Li Lithium 6.94 11 Na Sodium 22.99 19 K 39.10 28 Ni Nickel 58.69 37 Rb Rubidium 85.47 59 Pr 140.91 104 Rf (261) 42 Mo 95.94 101 Md (258) 32 Ge 72. For example, positive sodium ions and negative chloride ions react to form the neutral compound sodium chloride, commonly known as table salt.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
